Proliferation analysis of the growth plate after diaphyseal midshaft fracture by 5'-bromo-2'-deoxy-uridine.
"Both stimulative and inhibitory growth disturbances may occur after a fracture during the growth period. Chondrocyte proliferation [may lead] to overgrowth. This study investigates the effect of a fracture on the proliferation of chondrocytes at the nearby growth plate and its effect on the contra-lateral leg. Fifty-six 1-month-old Sprague-Dawley rats (weight, 100-120 g) were randomised to either an experimental or a control group. A closed mid-diaphyseal tibial fracture was produced in all animals of the experimental group using a standardised technique. On day 3, 10, 14 and 29 of the experiment, the rats were euthanised and their tibial growth plates were subjected to histological analysis. Proliferation of chondrocytes [increased] in the growth plates of broken bones during fracture healing. This proliferation peaked on day 3 post-fracture and then reduced gradually until day 29. No increase in the rate of proliferation was observed on the contra-lateral limbs of the animals in the experimental group. Following a diaphyseal fracture of the tibia, the growth plates located next to the fracture react with increased cell proliferation. This proliferation was not observed in the contra-lateral uninjured tibia."
What I don't get is if the fracture occurred at the middle of the shaft then shouldn't both growth plates be equally nearby? But during development the growth plate is a lot bigger. If stem cells were released from the trabecular bone they could travel through the blood into the growth plate and differentiate into chondrocytes whereas with a fully developed adult there's a lot smaller growth plate line to head into so the stem cells are less likely to make it there. Also, during development there's active genetic expression for various chondrocyte growth factors(which we re-activate by loading the articular cartilage during LSJL). In addition, during development things like the endiosteum are not fully formed and are not as developed hence it being easier for stem cells to get through to that growth plate.
Scientists would have to do tests on diaphyseal microfractures versus full on diaphyseal fractures to know which theory explained bone "overgrowth". If the bone overgrew during diaphyseal microfracture it would be due to a thinner endiosteum whereas if it only grew during full on diaphyseal fracture it would be due to stem cells traveling up and around.
Trabecular microfracture<-ideally to grow taller we want to see trabecular microfractures healing with endochondral ossification which we do see in a study on extracorporeal shockwave in rabbits. Specific study is Dose-dependent new bone formation by extracorporeal shock wave application on the intact femur of rabbits.
"Healing trabecular microfractures are a common feature in cancellous bone. These lesions, when observed in macerated cancellous bone slices, measure about 500 μm in diameter and surround fractures in trabeculae with microcallus. Whether microcallus is a structure acting primarily as a transient brace, preventing relative movement of the fragmented segments and enabling the trabecula to heal, or whether it is a permanent buttress reducing the stress on the fractured strut, preventing the healing process, is not known. Microfractures are the result of normal physical activity. Hence, the widespread occurrence of trabecular microfracture in cancellous bone implies that a reasonable rate of microfracture is physiologically tolerable. There are three putative effects for trabecular microfracture. One is that, in response to impulse loading, cancellous subchondral bone increases its rigidity due to osteosclerosis resulting from bone formed around microfractures. Another hypothesis is that, if sufficient trabecular microfractures occur, they will compromise the trabecular structure of the vertebra and the proximal femur leading to osteoporotic fracture. By inducing remodeling changes, microfractures have an effect on the maintenance of joint structure. There are two histological patterns for microfractures: an early stage, when actively forming woven bone is bridging the fracture; and a more common late stage, when woven bone is inactive. Femoral studies fail to demonstrate that an increasing number of healed or healing microfractures in osteoarthrosis causes the increase of bone in the head of femur. Only one study has reported a significant increase in the number of trabecular microfractures in osteoarthrotic femoral heads compared with normal controls. This significant increase was in patients taking antiinflammatory drugs. In osteoporotic fracture, sufficient trabecular microfracture may lead to femoral fracture. The bone loss in the vertebral bodies is by a loss of horizontal trabeculae. This loss reduces the resistance of vertical elements to deformation under load and creates the conditions for trabecular fracture. Coincident with this observation, microfracture is most prevalent on the vertical structure. The increase of microfractures with increasing age has three possible explanations: (l) the incidence of microfracture increases as trabeculae become thinner; (2) the incidence of microfracture is constant but the rate of healing decreases; or (3) these two factors combine to increase the number of microfractures. The occurrence of trabecular microfracture has been shown to correlate with factors such as physical activity, age, bone viability and remodeling potential, cancellous bone volume, bone mineral content, bone fatigue properties, and the direction of cancellous bone loading. As trabecular microfracture is not an event that initiates a pathological process, a number of important questions need to be addressed. Whatever the answers to these questions, trabecular microfracture is intimately linked to the nature of cancellous bone structure, and the conditions under which microfracture will compromise this structure are fundamental to the question of bone quality."
study is old so I didn't review it fully.
couldn't get full study.
"Compared to trabecular microfracture, the biomechanical consequences of the morphologically more subtle trabecular micro-damage are unclear but potentially important because of its higher incidence. A generic three-dimensional finite element model of the trabecular bone microstructure was used to investigate the relative biomechanical roles of these damage categories on reloading elastic modulus after simulated overloads to various strain levels. Microfractures of individual trabeculae were modeled using a maximum fracture strain criterion, for three values of fracture strain (2%, 8%, and 35%). Microdamage within the trabeculae was modeled using a strain-based modulus reduction rule based on cortical bone behavior. When combining the effects of both microdamage and microfracture, the model predicted reductions in apparent modulus upon reloading of over 60% at an applied apparent strain of 2%, in excellent agreement with previously reported experimental data. According to the model, up to 80% of the trabeculae developed microdamage at 2% apparent strain, and between 2% and 10% of the trabeculae were fractured, depending on which fracture strain was assumed. If microdamage could not occur but microfracture could, good agreement with the experimental data only resulted if the trabecular hard tissue had a fracture strain of 2%. However, a high number of fractures (10% of the trabeculae) would need to occur for this case, and this has not been observed in published damage morphology studies. We conclude therefore that if the damage behavior of trabecular hard tissue is similar to that of cortical bone, then extensive microdamage is primarily responsible for the large loss in apparent mechanical properties that can occur with overloading of trabecular bone."
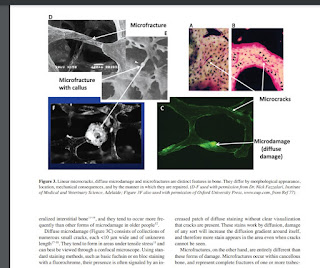
"Trabecular damage has been observed in the form of complete microfractures of whole trabeculae and microdamage within trabeculae, including longitudinal and transverse microcracks, diffuse damage, and cross- hatched patterns "
"There are a at least three distinct
varieties of “microdamage,” which can be identified as linear
microcracks, diffuse microdamage, and microfractures"
Above is a microfracture healing via endochondral ossification.
"Linear microcracks and diffuse damage both repair through normal coupled remodeling processes, although signaling for remodeling may differ between them with linear microcracks having greater potential to initiate a repair response. They are removed by resorption, and new bone is laid down where the damage once was. Microfractures, on the other hand, are repaired through normal fracture healing mechanisms which involve endochondral ossification"
look up Teriparatide for stem cells..
ReplyDelete